What are cancer clinical trials, and why are they so important?
Cancer’s impact on Minnesotans is no secret. Nearly half of all Minnesotans will be diagnosed with a potentially life-threatening cancer during their lifetimes. One out of four Minnesotans die of cancer. And cancer is the leading cause of death in our state. That’s why it is critical that Minnesota’s healthcare providers have access to cutting-edge cancer research—like the research our team at the Masonic Cancer Center (MCC), University of Minnesota conducts every day.
No one is working harder than our team of experts at MCC to advance the science of cancer care. Every day, patients benefit from working with our specialized team of doctors, nurses, researchers, and healthcare professionals committed to providing life-saving care. Advances in that life-saving care are the direct result of new ideas and approaches developed through research at MCC—including clinical trials.
Clinical trials are studies of new medicines, procedures, and other types of treatments in people that doctors and researchers use to develop new methods of treating serious diseases, like cancer. But there are many aspects of the clinical trials process that are not widely talked about or well known. Let’s take a look at some of the most common questions and/or concerns about clinical trials and their significance to cancer research and care.
Why do we need clinical trials?
Clinical trials help inform our understanding of cancer and improve prevention, diagnosis, treatment, and care—they show us what works and what doesn’t and help build the future of medicine. They are the best way to answer critical questions about cancer, from preventing it in the first place, to finding and diagnosing it, treating it, and managing its symptoms and the side effects of its treatments.
Today’s clinical trials often become tomorrow’s new standard of care, boosting many patients' quality of life now and helping ensure that future patients have continuously higher standards of care for years to come. This progress could not occur without the people who take part in clinical trials—and the people who get patients connected to them: doctors, nurses, and other healthcare professionals who work to ensure that anyone who needs access to a clinical trial can be part of one.
There are two main types of clinical trials: therapeutic trials and non-therapeutic trials. Therapeutic trials enroll patients and provide a specific treatment to the patients to study that treatment’s impact on cancer. Non-therapeutic trials do not provide a treatment to patients but instead study important factors which help advance the understanding of cancer and its impact—from prevention and screening to survivorship. For example, some non-therapeutic studies collect tissue specimens to examine the cellular structure of a cancer tumor. Other studies track epidemiological information such as the long-term health effects of chemotherapy. Stay tuned for another blog dedicated to non-therapeutic clinical trials, coming soon.
What are the phases of clinical trials?
Clinical trials are done in stages, called phases. Before any prospective treatment is used with people in clinical trials, researchers work for many years to understand that treatment’s effects on cancer cells, both in laboratories and on animals, and they try to figure out any side effects that particular treatment may cause.
If pre-clinical research for a treatment is completed and the treatment still seems promising, researchers will ask the U.S. Food and Drug Administration (FDA) to give permission for that treatment to be studied in humans—this is called an investigational new drug, or IND, application. The IND application must contain specific information, and whoever is conducting the research must commit to getting informed consent—or written permission—from everyone participating in the clinical trial. Researchers must also commit to having the study reviewed by a special institutional review board (IRB) and to following all the rules required for human subjects research.
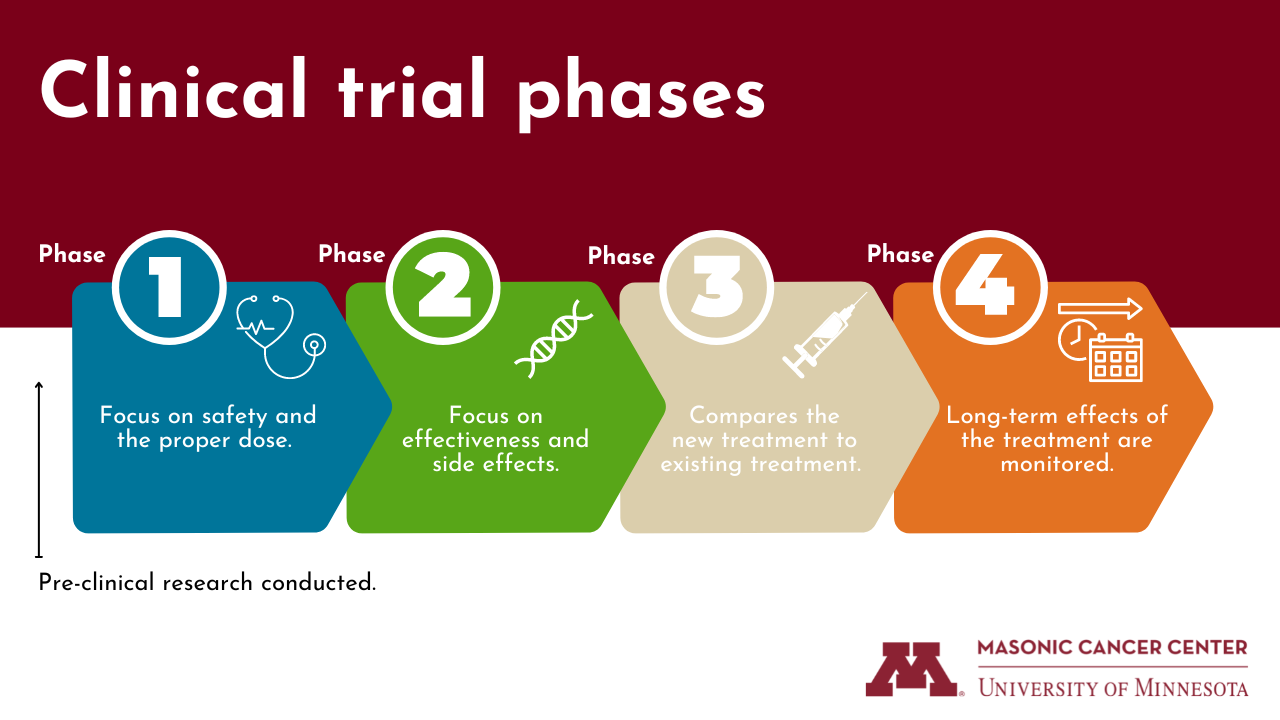
From there, a clinical trial can go through a series of phases—although not all trials go through every phase:
-
Phase 0—exploring if and how a new drug may work. The purpose of this phase is to help speed up and streamline the drug approval process. Although not widely used and not a required phase, phase 0 studies may help researchers find out if the drugs do what they’re expected to do and save them time and money on future phases. Phase 0 studies use only a few small doses of a new drug in a few people. They might test whether the drug reaches the tumor, how the drug acts in the human body, and how cancer cells in the human body respond to the drug.
-
Phase I—is the new treatment safe? Phase I trials are usually the first that involve people. In this phase, the new treatment is tested to see if it's safe and to find out what dose works the best without causing severe side effects.
-
Phase II—does the treatment work? Phase II trials test the effectiveness and side effects of the treatment and if it works in certain types of cancer.
-
Phase III—is it better than what's already available? Phase III trials compare the safety and effectiveness of the new treatment against the current standard treatment—one of the best options currently being used to treat a specific type of cancer. In this phase, researchers focus on how side effects for each type of treatment (the new treatment and the current standard) might be different.
-
Phase IV—what else do we need to know? New treatments are often watched over a long period of time in phase IV trials. That's because, even after testing a new medicine on thousands of people, all the effects of that treatment may still not be known. Phase IV trials give researchers the chance to study a treatment over several years.
How do we protect people participating in clinical trials?
There have been many moments throughout history when the medical and research community has not properly served people, leaving patients and their loved ones with rightful concerns about what the clinical trials process is like and what safeguards will be in place throughout it.
Clinical trial participants are a vital part of cancer research, and there are federal rules in place to help ensure the safety and ethics of clinical trials—whether therapeutic or non-therapeutic.
For patients considering cancer clinical trials, common myths about them can often be a barrier to participating. Thanks to a collaboration between the MCC Community Advisory Board and the University of Minnesota Hubbard School of Journalism, these videos help break down some of the most common myths about clinical trials.
Every trial or research study has a person in charge—usually a doctor—who is called the principal investigator (PI). The PI prepares and carries out the clinical trial or research study’s plan, also called a protocol, and then analyzes the data and reports the results of the trial or study. The protocol for a clinical trial explains what will be done during the trial and contains specific information on the reason for doing the trial, who can join it, drugs or treatments that will be given, types of medical tests that will be done, and types of information that will be collected that helps a doctor decide if that particular treatment is right for a patient.
In addition to this information being accessible to potential trial participants, all clinical trials are bound by three values:
- Respect. All people should be respected—they have the right to choose what treatments they get.
- Beneficence. The welfare of research participants is the main goal. Clinical trials must protect people from harm by giving the most benefit with the least amount of risks possible.
- Justice. All clinical trials must ensure that all participants share the benefits and burdens equally.
How do people participate in clinical trials?
Every clinical trial has different patient requirements, possible benefits, and risks. Physicians and oncologists will speak with their patients to discuss all possible treatment opportunities, including clinical trials. And all patients will be told about any known risks from a clinical trial or other treatment opportunity before it begins.
Curious which clinical trials are available at the Masonic Cancer Center and throughout Minnesota? Find all available clinical trials at MCC and filter them by cancer type. The Minnesota Cancer Clinical Trials Network also provides a full list of open trials across the network’s locations in Minnesota.
For more detailed information on clinical trials that may be available, call the Masonic Cancer Center Nurse Navigator Line at 612-624-2620. The line is staffed Monday through Friday, 8 a.m. to 4:30 p.m.
How is MCC improving equitable access to cancer clinical trials for all Minnesotans?
It is critical that all Minnesotans facing a cancer journey are able to access information and resources about clinical trials as well as participate in them in their own communities. That’s where the Minnesota Cancer Clinical Trials Network (MNCCTN) comes in, led by MCC in partnership with five healthcare organizations in Minnesota: Essentia Health, M Health Fairview, Mayo Clinic Health System, Metro-Minnesota Community Oncology Research Consortium (MMCORC), and Sanford Health.
MNCCTN’s trials originate from our state’s two National Cancer Institute (NCI)-designated Comprehensive Cancer Centers, the Masonic Cancer Center and Mayo Clinic Cancer Center, as well as from the Hormel Institute in Austin, Minnesota. MNCCTN brings together healthcare organizations from across the state to improve cancer outcomes for all Minnesotans through greater access to clinical trials. This network consists of 22 site locations across Minnesota that offer cancer clinical trials—and more are being added each year.
MNCCTN exists specifically to bring cancer clinical trials directly to those living in Greater Minnesota, as many community clinics do not have the resources to establish the necessary equipment, staff, and training to conduct clinical trials. This breaks down barriers like:
- Distance. Prior to MNCCTN, clinical trials were available in limited locations.
- Time. Traveling to these few locations required significant time away from work and family.
- Costs. Traveling incurs many indirect costs to patients such as fuel, lodging, and meals.
- Comfort. A familiar setting, doctors, and being "at home" reduces the emotional burden on participants.
Plus, having access to a larger portion of the state's population also helps researchers move successful treatments through the clinical trials process more quickly, expanding knowledge and advancing the time in which new treatments and cures are available to the general public.
All of us at the Masonic Cancer Center recognize and honor the commitment our community of Minnesotans makes in participating in clinical trials that advance the science of cancer care—whether your life has touched a cancer journey directly or you support us financially or by sharing the word about the innovative research and treatments our MCC team is part of. We are committed to continuing to show up every day to ensure all patients and their loved ones who work with our specialized team of doctors, nurses, researchers, and healthcare professionals receive life-saving care and support through all stages of a cancer journey.
Still have questions? Join us at our next Fireside Chat to get answers straight from the experts! We're hosting Nisha Patel, engagement director for global demographics and diversity at GSK, and Dr. Jeff Miller, deputy director of the Masonic Cancer Center and hematologist and oncologist with MCC tomorrow at 1 p.m. Central to discuss the drug development process, the use of clinical trials in healthcare, barriers to clinical trials participation, and more.
