All About Cancer Clinical Trials: Trial Phases
One of the most common types of cancer clinical trials are treatment trials. Treatment trials study whether a new treatment is safe, effective, and superior to existing treatments. Treatment trials often study new drugs. However, other treatment trials can study devices, surgical procedures, radiation therapy, or other treatment methods. Treatment trials are further categorized by stage, or phase. There are four main clinical trial phases. However, before these phases begin, new treatments undergo preclinical studies.
Preclinical Studies
These are also referred to as laboratory studies. In preclinical studies, treatments are tested for safety, but not on people. Before a treatment can be given to people in a clinical trial researchers must have evidence that it could work and be safe for people. Treatments can be tested using animals or cell models. In cell models, cancer cells are grown in a lab in a dish or test tube. Then, a treatment is tested on these cells to see how they react. Next, the treatment may be given to animals to make sure it is likely the treatment would be safe in other living animals like humans.
IND & Phase 0 Trials
If cell and animal models indicate that the treatment is promising, researchers must submit the treatment to the Food & Drug Administration (FDA) to get permission to move on to Phase I trials before the treatment can be tested on people. This process is called the investigational new drug (IND) application. If the IND is approved by the FDA, then the treatment can move on to the next phases.
Phase 0 trials are uncommon but may occur for some treatments. Phase 0 trials test low doses of a drug to a very small number of people (fewer than 15) to learn more about a treatment and how it may work in people. Phase 0 trials are not a required phase in the clinical trial process.
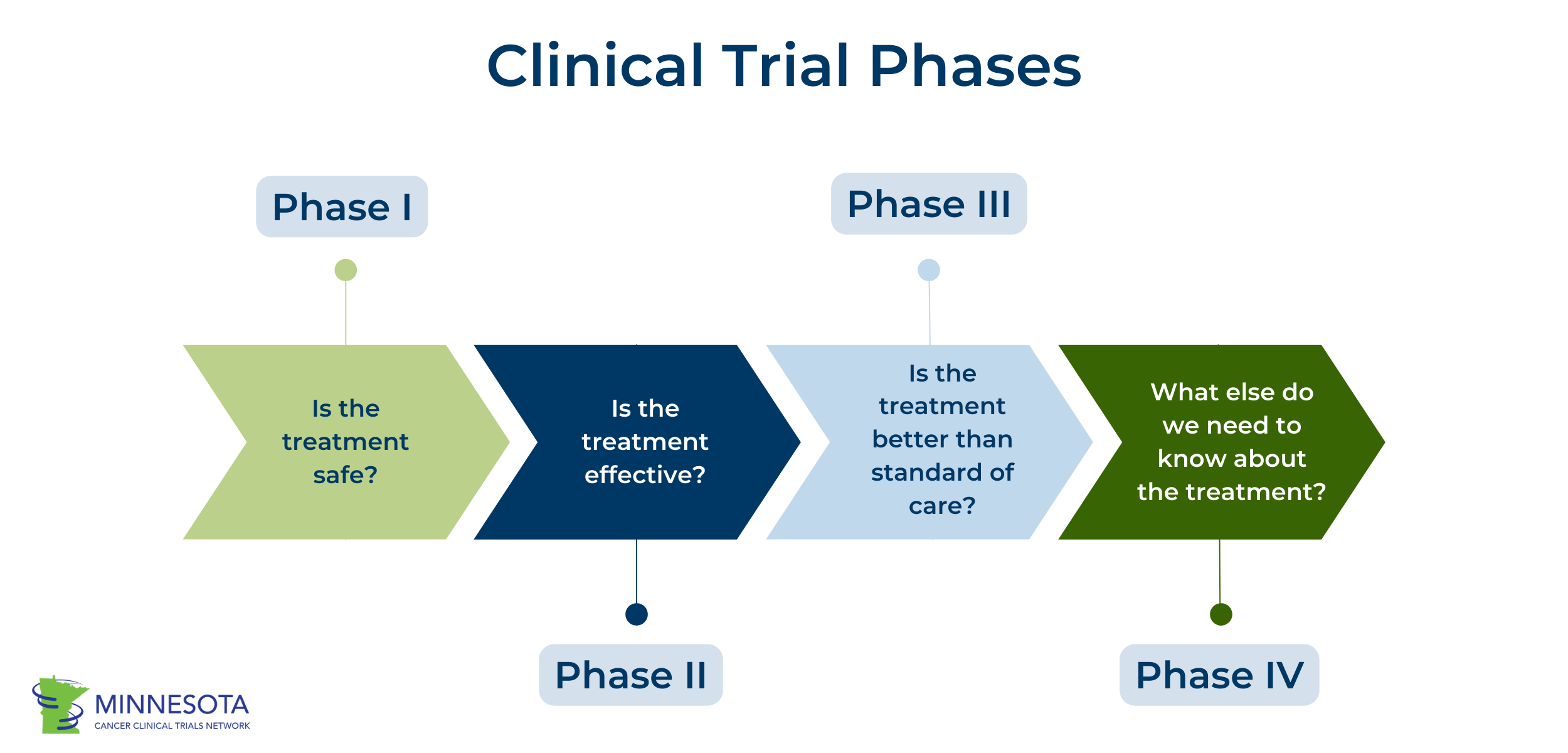
Phase I Trials
In Phase I trials, researchers test a new drug or other type of treatment on people, usually for the first time. These studies include a small group of people, usually 20-50 people. The focus of Phase I studies is to learn about drug or treatment safety and identify side effects. Because this is often the first time a drug or other treatment will be tested on people, Phase I trials carry the most risk. Sometimes researchers will start with low doses and gradually increase them and monitor for safety and side effects. If the treatment is shown to be safe, it can move on to Phase II trials.
Phase II Trials
Phase II trials test the drug or treatment on a larger group of people (around 100 or more people). The goal of Phase II trials is to determine how effective the drug or treatment is for certain types of cancer. Effectiveness depends on the type of cancer and treatment. It might mean that a tumor shrinks or stays the same. It could also mean that the participant is cancer-free after treatment, or that a participant lives longer. Effectiveness could also mean improved quality of life. Phase II trials also further study safety. If shown to be effective, the treatment can move on to Phase III trials.
Phase III Trials
Phase III trials include an even larger group of people (hundreds to several thousands of people). These studies further study treatment effectiveness, safety, and side effects. Phase III trials also typically compare the new drug or treatment with standard of care, similar treatments, or a placebo. A placebo is a treatment that doesn’t have any effect. Cancer trials don’t use placebos very often and instead compare a new treatment to standard current treatments (standard of care). The study will determine if the new drug is more effective or beneficial than other treatments. In these studies, participants are typically assigned to a group randomly, and this group determines which treatment they will receive. This is called randomization. Phase III trials also collect information about how to use the treatment safely. If a treatment is shown to be safe and more effective than current treatments, researchers can submit the drug to the FDA for approval. This is called a new drug application (NDA). The FDA will review the previous clinical trials and evidence and determine whether or not to approve the new treatment. If approved, the drug can be made available to the public and become standard of care. If the treatment is not approved, researchers may need to provide more information or conduct more studies.
Phase IV Trials
Phase IV trials occur after a drug or treatment is approved by the FDA and made available to the public. Also referred to as post-marketing surveillance studies, Phase IV trials are conducted for a long period of time after FDA approval. Phase IV trials track a treatment’s safety, describe risks and benefits, and determine optimal use of the treatment. These studies may involve thousands of people.
Learn More About Cancer Clinical Trials
Interested in learning more about cancer clinical trials and the basics of participation? Check out MNCCTN’s previous blog posts in the “All About Cancer Clinical Trials” series!
Types of Cancer Clinical Trials
